
In the second part of the chapter, it is made clear that dilution is usually made by reducing the mass of the drug present in the original stock solution while keeping the final volume of the diluted solutions constant. 
The concept of the dilution factor is introduced to cope with problems that deal with the construction of calibration curves for quantitative drug analysis in dosage forms and biological fluids. This same equation is then used to calculate the concentration of drugs in pharmaceutical products prepared by diluting a known amount of drug with other excipients or to calculate concentration of drugs in patient’s dose. 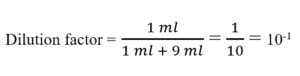
The concept of dilution is first explained using a mass balance equation that was generated based on the fact that the mass of a substance is preserved during dilution processes implicated in compounded drug preparations. This chapter is essentially divided in two parts, that is, dilutions in drug compounding and dilutions in drug analysis, which are more useful to individuals engaged in research projects and laboratory work.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
